Theory to Action: Applying CRISPR in Research
Traditional Gene Knockout
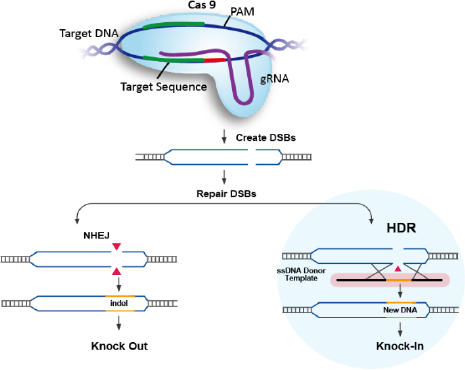
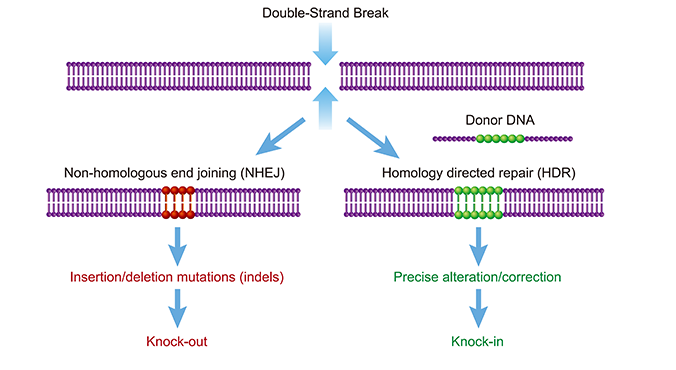
As described above, the CRISPR-Cas complex generates a targeted double-strand
break,
creating an opportunity for gene editing.
Gene knockout (KO) experiments exploit flaws in the cell’s native repair mechanism,
non-homologous end
joining (NHEJ). Imperfectly repaired genes with insertions or deletions of base pairs will
result in
frameshift mutations, rendering the gene and its corresponding protein non-functional. KO
guides are
algorithmically designed and ranked for both specificity (low homology with any other
genomic sequence)
and functionality (targeting a portion of the gene most likely to result in functional
protein KO).
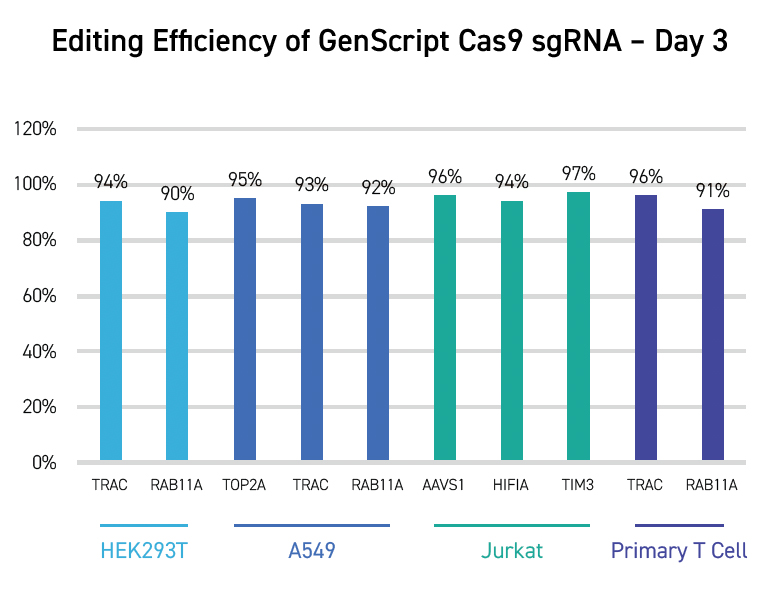
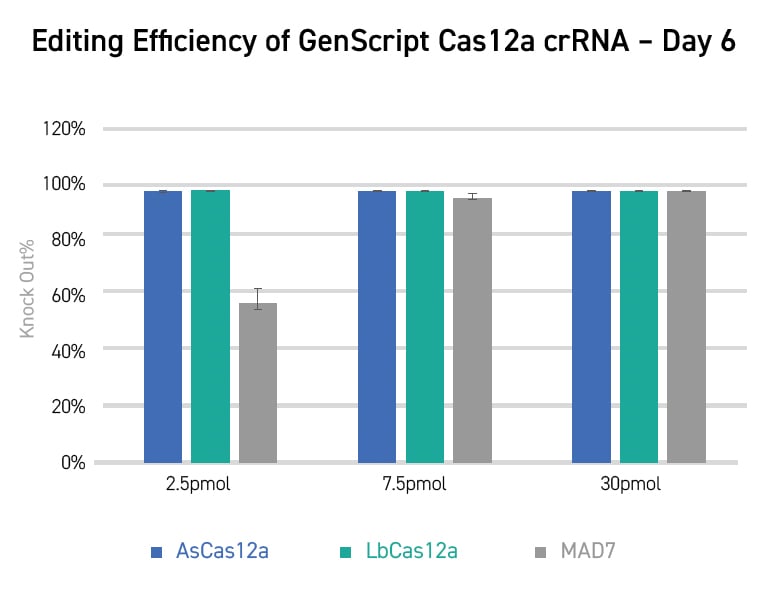
CRISPR KO can be applied in many research scenarios, from basic screening, functional
studies of a gene,
bioengineering, agricultural biotechnology, to developing therapeutic gene and cell
therapeutic drugs.
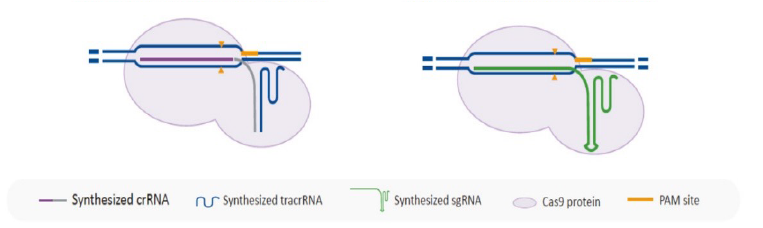
Increasing CRISPR/Cas9 Editing Specificity
CRISPR Cas9 based gene knockout relies on the specificity of both the sgRNA and
the Cas9
protein. Poorly designed sgRNA and traditional wildtype Cas9 would lead to editing at
undesired genome
locations, which are considered as off-target edits. Many Cas9 variants have been developed
for enhanced
specificity. For example, the eSpCas9(1.1), also referred to as SpCas9
(K848A/K1003A/R1060A),
structurally engineered from Feng Zhang lab contains alanine mutations that weaken the
bounding between
the HNH/RuvC groove and the none targeting DNA strand, reducing off-target effects by over
10-fold while
maintaining robust on-target genome editing efficiency (Slaymaker et al. 2016).
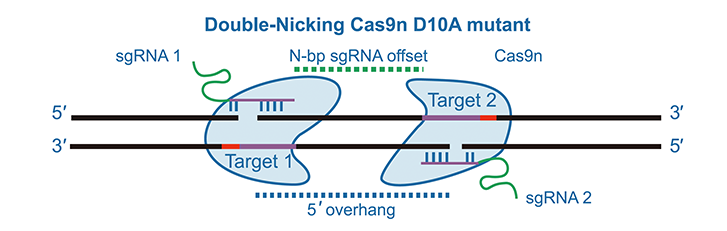
Researchers have also engineered CRISPR Cas variants, such as Cas9 nickases, to
“nick” a single DNA strand instead of creating double-stranded DNA breaks, enhancing editing
precision. Two individual nickases targeting opposite DNA strands
are
necessary to generate a break site for gene editing, which significantly increases targeting
specificity. For example, SpCas9 nickase (Cas9n D10A) contains a mutation allowing the
endonuclease to
create single-strand nicks, as opposed to DSBs. Pairing two opposite facing gRNA sequences
with SpCas9
nickase can be an efficient method of gene editing that prevents unwanted indels from
forming.
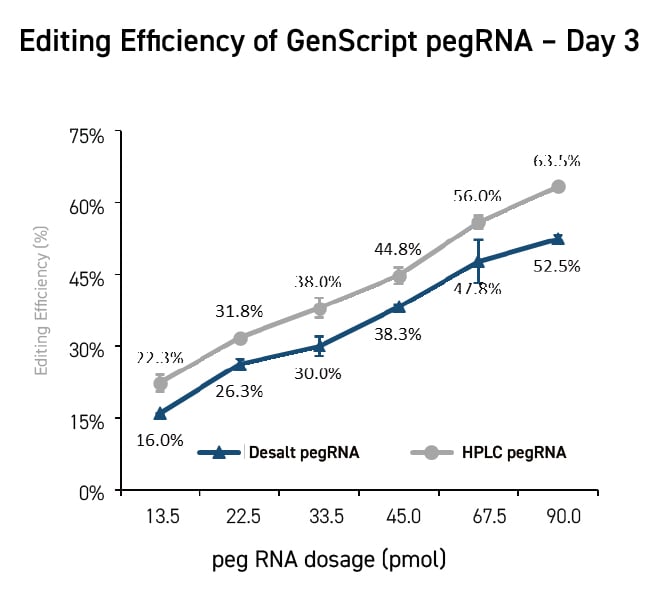
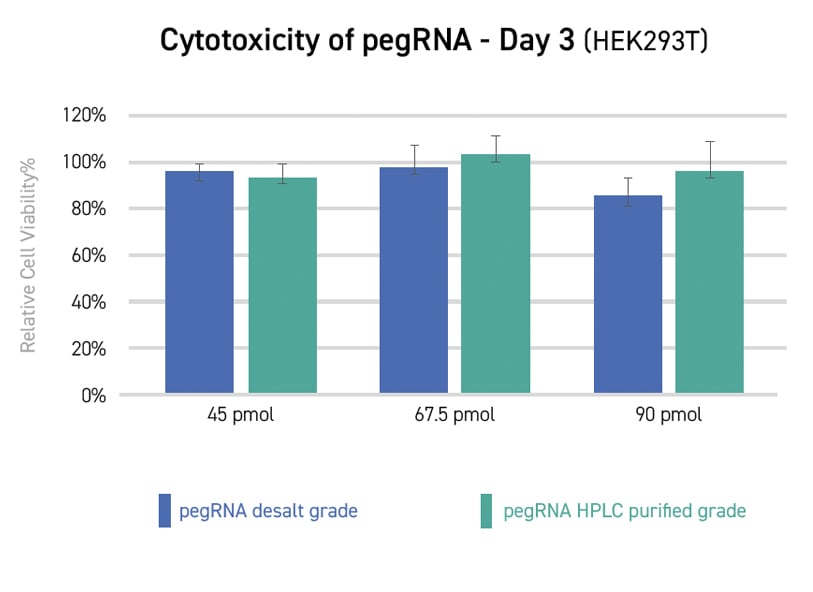
Novel Editing Technologies Without DSBs (Prime Editing/Base
Editing)
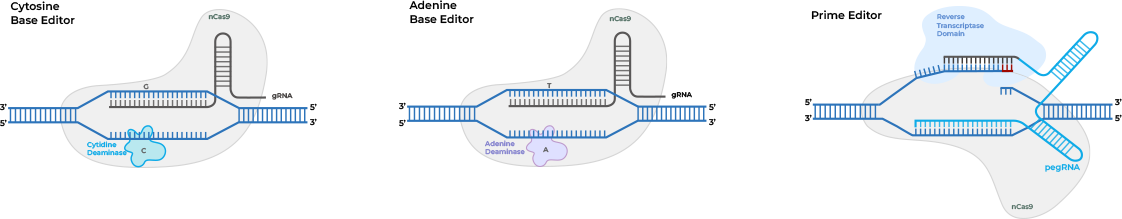
Cas9 nickases also could enable novel single-strand CRISPR gene editing
techniques that
avoid the potential for unintended genomic changes posed by double-strand DNA breaks.
Prime
editing
utilizes a Cas nickase protein fused to a reverse transcriptase (RT) to write a new sequence
into a
target DNA site directly. Prime editing guide RNAs (pegRNA) contain both a DNA targeting
sequence and an
RT template so that the pegRNA-dCas9 nickase complex can identify the target site, cut a
single strand,
and write the new sequence. Multiple versions of the prime editing system have been
developed to improve
editing efficiency.
Base editing utilizes a Cas9 nickase, or alternatively a catalytically dead
Cas9
(dCas9) which only
binds to target DNA without cutting, fused to a nucleobase deaminase enzyme and a DNA
glycosylase
inhibitor to make targeted point mutations. For example, cytosine base editors (CBEs) and
adenine base
editors (ABEs), by fusing Cas9 nickase or dCas9 to a cytidine deaminase like APOBEC or
adenine DNA
deaminases, can convert C to T (or G to A) and A to G (or T to C).
Traditional Gene Knockin
Gene knock-ins have various applications in biotechnology, including disease
modeling and
gene and cell therapy applications. CRISPR based gene insertion relies on a pathway
different than NHEJ,
it is called Homology directed repair (HDR) pathway. HDR process is very precise allowing
for accurate
gene knockin, but HDR occurs in less frequency compared to NHEJ leading to less editing
efficiency
compared to knockout. In these experiments, the Cas9 cut site plays a critical role, as it
determines where the repair process occurs. Successful HDR also requires the introduction of
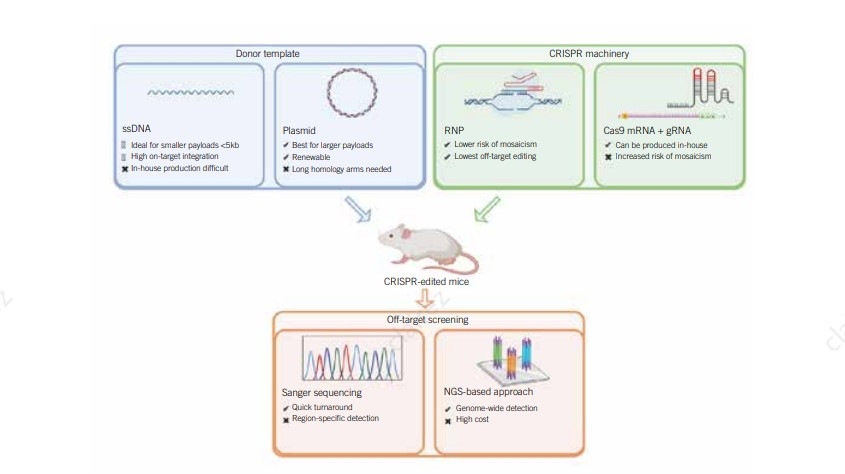
Donor DNA HDR templates, which can be single- or double-stranded DNA designed with
overlapping homology arms to the specific Cas9 cut site for precise gene integration.
To increase the efficiency of HDR, experiment optimization by testing different
template
format, delivery approaches, culture conditions, as well as using small molecules for cell
cycle
synchronization, enhance HDR or inhibit NHEJ have been used to overcome this challenge.
Read more on HDR
template
formats available and design.
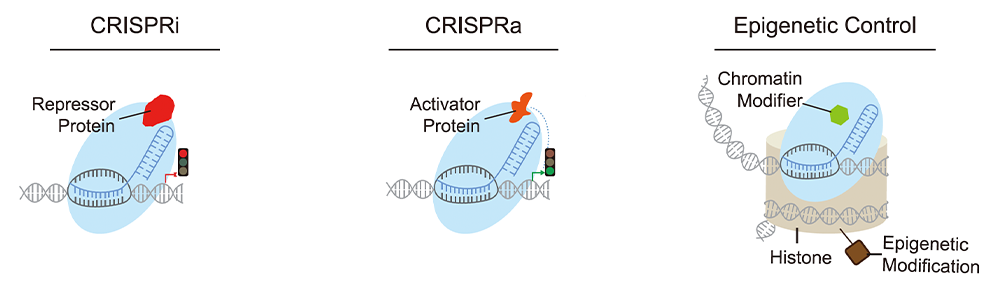
CRISPR Transcriptional Regulation
Additional Cas9 variants have been developed for transcriptional regulation of
gene
expression. Rather than editing DNA, these deactivated Cas9 proteins simply bind the target
site and
activate or inhibit expression.
CRISPR activation (CRISPRa) systems utilize a dCas9 fused to transcriptional
activators to
up regulate endogenous gene expression, whereas CRISPR interference (CRISPRi) systems use
dCas9 fused to
transcriptional repressors to downregulate gene expression. Multiple CRISPRa and CRISPRi
systems have
been developed using different activators or repressors. CRISPRa
gRNAs for the SAM system are currently
available in plasmid formats.
Researchers have also recently developed a Cas fusion protein, CRISPRoff, that
can
epigentically modify and silence genes without editing the underlying genome. The epigenetic
memory
persists through cell differentiation and is heritably passed to future cell generations.
Gene activity
can be re-activated using a different fusion protein called CRISPRon.
| Technique |
Components Required |
Common Experiments |
Common Applications |
| CRISPRa |
CRISPRa guide RNA + dCas9-SAM
(or other) |
Overexpression screening and hit analysis |
Drug discovery |
| CRISPRi |
CRISPRi guide RNA + dCas9-KRAB
(or other) |
Transient loss-of-function, siRNA validation |
Drug discovery |
CRISPRoff/
CRISPRon |
CRISPRoff guide RNA + CRISPRoff |
Heritable gene silencing without DNA editing |
Gene therapy |