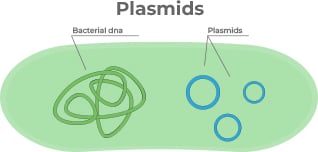
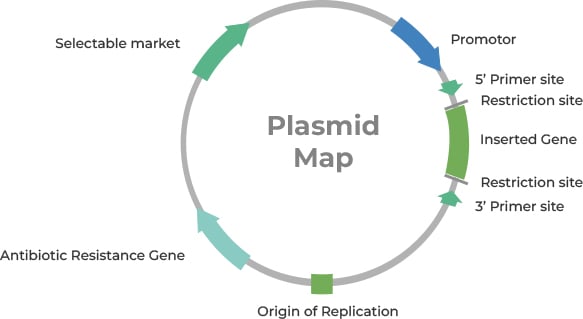
Having delved into the intricacies of plasmid vector components, it is
now clear that the success of these vectors is not only determined by their
intrinsic design. Even the most elegantly constructed plasmid requires an optimal
environment to manifest its potential. This leads us to the pivotal next phase:
propagating these plasmids in host strains. While complex organisms such as mammals,
yeast, and fungi serve as host choices for niche applications like protein
production, bacteria—especially Escherichia coli (E. coli)—remain the
preferred
choice due to their rapid growth, ease of cultivation, and cost-effectiveness.
In this week’s digest, we dive deep into the background, types, and
applications of E. coli strains.
Introduction to Host Strains
Background:
At their core, host strains are living organisms or cells employed to
introduce,
replicate, and express foreign DNA. This typically involves the introduction of plasmids or
other DNA constructs into the host for a myriad of applications, ranging from plasmid
propagation and protein production to genetic studies and more.
Strains vs Species:
A strain represents a subset within a species distinguished by unique
characteristics. While every member of a strain belongs to a species, not every member of a
species belongs to a specific strain.
Common Uses of Host Strains:
- Gene/Protein Expression:
Introducing a desired gene into a host strain to produce a specific protein or enzyme.
- DNA Cloning: Replicating DNA
in host strains to generate multiple copies.
- Metabolic Engineering:
Modifying host strains to synthesize chemicals, drugs, or biofuels.
- Probiotic Research:
Engineering beneficial bacteria for improved human health.
Types of Host Strains:
- Bacteria: Escherichia
coli (E. coli) is the most widely used bacterial host.
- Yeast: Saccharomyces
cerevisiae (Baker’s
yeast) used in baking and brewing. Additionally, it is also employed for protein
production.
- Fungi: Aspergillus species,
used in enzyme production.
- Mammalian Cells: Chinese
Hamster Ovary (CHO) cells are used for protein production
Evolution of Host Strains:
With scientific advancements, the selection and optimization of host strains
have continually evolved. While natural strains were initially harnessed as
hosts, they posed challenges related to yield, safety, and genetic stability. Mutagenesis,
selection, and genetic engineering paved the way for optimized strains tailored for
laboratory and industrial utility.
Introduction to Escherichia coli Host Strains
Escherichia coli, often dubbed E. coli, is a rod-shaped bacterium
indigenous to
the intestines of warm-blooded creatures. Most strains are benign and symbiotically coexist
with their hosts, but a few can trigger ailments. In nature, E. colichampions gut
health
and aids digestion. By outcompeting pathogenic counterparts, it bolsters natural immunity.
Nonetheless, certain pathogenic strains, when consumed, can induce ailments like food
poisoning. Beyond its natural existence, E. colihas carved a niche as a go-to model
organism in scientific exploration and biotechnology.
E. coli as a Model Organism:
- Genetic Simplicity: E.
coli's singular,
circular chromosome ensures it is genetically less intricate than eukaryotic organisms,
streamlining genetic studies.
- Rapid Growth: E.
coli can double its
population approximately every 20 minutes under optimal conditions, making it an
excellent host for DNA propagation and protein production.
- Affordability: It has basic
and cost-effective growth requirements.
- Extensive Research: E.
coli is one of the
most extensively researched organisms, offering detailed insights into its molecular
biology. Notably, its genome was fully sequenced as early as 1997.
Advantages of Using E. coli Host Strains:
- High Transformation
Efficiency: These strains have a high affinity for foreign DNA, which makes
them particularly suited for cloning and other genetic engineering endeavors.
- Yield Potential: They can
yield large amounts of proteins or nucleic acids in relatively short time frames.
- Diverse Genetic Tools: A vast
assortment of plasmids, phages, and other genetic tools have been specifically designed
for E. coli.
Types of Laboratory E. coli Strains:
Strains of E. coli derived in the laboratory have been
intentionally
selected or modified to meet experimental needs. Over the years, scientists have
identified and engineered various E. coli strains tailored for specific
tasks, such
as cloning and protein expression. Each of these strains possesses unique mutations
and is a product of advanced genetic engineering.
Cloning Strains
E. coli cloning strains are genetically tailored to maximize
efficiency
and stability during the introduction of exogenous DNA fragments. "Importantly,
these strains typically have mutations that prevent unwanted DNA degradation and
recombination, ensuring the effective uptake and amplification of the introduced
genetic material.
Table 1. Common E coli Strains for Cloning
| Strain |
Genotype |
Applications |
| DH5α |
F–, φ80dlacZΔM15, Δ(lacZYA-argF)U169,recA1, endA1, hsdR17(rK–, mK+), phoA,
supE44, λ–thi-1, gyrA96, relA1 |
General cloning applications, Blue/White Screening |
| Top10 |
F–mcrA, Δ(mrr-hsdRMS-mcrBC), φ80lacZΔM15, ΔlacX74, recA1, araD139,
Δ(ara-leu)7697, galU, galK, λ–rpsL(StrR), endA1, nupG |
General cloning applications, Blue/White Screening, High transformation
efficiency |
| Stbl3 |
F–mcrB, mrr, hsdS20(rB–, mB–), recA13, supE44, ara-14, galK2, lacY1, proA2,
rpsL20(StrR), xyl-5, λ–leu, mtl-1 |
Ideal for cloning vectors with direct repeats (LTRs in lentiviral plasmids)
|
Note : F-: Deletion of F factor; F+: Autonomous F factor, does not carry
any genetically identifiable chromosomal fragment.
Table 2. Gene Symbols and Meanings
| Gene Symbol |
Meaning |
Annotation |
| Δ |
Deletion |
Deletion mutation is represented by "Δ", followed by the name of the deleted
gene and its allele number. For example, Δ(lac-proAB) represents
the deletion of the lac-proAB gene. |
| : |
Break |
: indicates that the gene before ":" is broken. |
| :: |
Insertion |
The gene before "::" is interrupted due to the insertion of the gene after
"::" |
| + |
Dominant or resistant |
If representing resistance, "+" can also be replaced by "r". |
| - |
Recessive or sensitive, non-resistant |
If representing sensitivity to a certain antibiotic, it is indicated by a
superscript "-" |
| () |
|
The gene in () represents the location of deletion or variation. |
| Φ |
Fusion |
Φ(ara-lac) represents the fusion of ara and lac
genes into a new gene |
Table 3. Common mutations for E coli Cloning Strains
| Gene |
Impact |
Found in strains |
| ΔendA1 |
Reduces intracellular endonuclease activity, resulting in high-quality
plasmid DNA by minimizing nonspecific digestion |
DH5α, Top10 |
| recA1 |
Reduces homologous recombination, which provides stability to cloned DNA.
|
DH5α, Top10, Stbl3 |
| ΔlacZΔM15 |
Allows for the blue-white screening method, facilitating the identification
of clones with vector insertions based on color. |
DH5α, Top10 |
| Δ(mcrA)183 |
Renders the strain incapable of restricting methylated DNA, making them more
receptive to DNA from various sources. |
Top10, Stbl3 |
| relA1 |
Affects the stringent response, which can influence the stability of certain
cloned sequences. |
Stbl3 |
Expression Strains
Expression strains are
specialized variants of E. coli that are optimized for producing proteins
from
introduced genes. They harbor specific genetic elements and mutations that enhance
protein synthesis, folding, and post-translational modifications.
Table 4. Common E coli Strains for Expression
| Strain |
Genotype |
Applications |
| BL21 (DE3) |
F–ompT, hsdSB, (rB–, mB–) gal dcm (DE3) |
General protein over-expression |
| Rosetta (DE3) |
F-ompT, hsdSB(rB- mB-) gal, dcm, (DE3), pRARE, (CamR) |
Optimized for expressing eukaryotic proteins that have codons rarely used in
E. coli, enhancing protein yield and solubility.
|
| Origami (DE3) |
F-ompT, hsdSB(rB- mB-), gal, dcm, lacY1 ahpC, (DE3), gor522::Tn10, trxB,
(KanR, TetR) |
Ideal for proteins that require disulfide bonds for proper folding,
enhancing the proper production and functionality of such proteins in E.
coli. |
| C41(DE3) and C43(DE3) |
F –, ompT, hsdSB, (rB- mB-), gal, dcm, (DE3) |
Engineered to improve the expression of membrane proteins or other toxic
proteins that are difficult to produce in standard BL21(DE3) cells. |
Table 5. Common mutations for E coli Expression Strains
| Gene |
Impact |
Found in strains |
| hsdR, hsdM, hsdS |
These mutations are part of the R-M (restriction-modification) system, which
restricts foreign DNA. Mutations in these genes reduce this restriction
activity, preventing degradation of introduced DNA. |
BL21(DE3), Rosetta (DE3), C41(DE3), C43(DE3), Origami (DE3) |
| T7 RNA polymerase gene (in the lambda DE3 lysogen) |
Provides the machinery for strong and controlled protein expression under
the T7 promoter. |
BL21(DE3), Rosetta (DE3), C41(DE3), C43(DE3), Origami (DE3) |
| pRARE |
Supplies tRNAs for seven rare codons in E. coli, facilitating the
expression
of eukaryotic proteins with such codons. |
Rosetta (DE3) |
| trxB gor |
Promotes disulfide bond formation in the cytoplasm, crucial for proper
folding of certain proteins. |
Origami (DE3) |
| rne131 |
Reduces RNase E activity, leading to increased mRNA stability and
potentially higher protein yields. |
Found in specialized strains |
Considerations when Selecting the Right Strain:
- Purpose of Experiment
- a. DNA Cloning: Consider strains with high transformation efficiency, such as
DH5α or XL1-Blue.
- b. Protein Expression: BL21 and its derivatives are popular choices.
- c. Reporter Assay: Strains such as XL1-Blue are suitable for blue-white
screening.
- Genomic Stability: If you are
working with
large or unstable DNA sequences, opt for strains that reduce recombination (e.g.,
strains with a recA mutation).
- Plasmid Compatibility: Ensure
the chosen
strain can stably maintain and propagate the plasmid you intend to use. This is
particularly important if you're working with a less common plasmid type. Be aware that
some strains contain natural resistances that make them resistant to certain
antibiotics.
- Growth Rate and Yield: Certain strains
provide faster
growth rates or yield higher amounts of proteins or nucleic acids. These qualities can
be crucial for large-scale productions or when time is of the essence.
- Endotoxin Levels: When producing
therapeutic proteins or
in other contexts where endotoxin levels are crucial, consider strains engineered to
produce lower levels of endotoxins.
- Sensitivity to Conditions or Compounds: If
your study
entails subjecting bacteria to specific conditions (e.g., temperature changes,
chemicals), it's essential to select a strain that can withstand those conditions.
Conclusion
Selecting the right E. coli strain is crucial for successful research
or
production activities, requiring careful consideration of factors like experiment type,
genomic stability, and plasmid compatibility. This choice, when made appropriately, can
significantly enhance outcomes, showcasing E. coli's vast utility in biotech. Given
the
complexity of selecting the right E. coli strain, seeking expert guidance can be
invaluable.
At GenScript, our plasmid preparation services
are tailored to diverse challenges, be it
plasmids with repeated ITRs, high GC content, unstable Poly-A sequences, or extended lengths
(>200kb); we have specialized host strains to cater to your research demands.
Additional reads
- Casali N (2003) Escherichia coli Host Strains. In: Casali
N, Preston A (editor), E. coli Plasmid Vectors: Methods and Applications.
Totowa: Humana Press, pp 27–48.











































































Notable Publications
Publish in High Impact Factor Journals
Rising Citation Count