
Fermentor

Fermentor
 IVD Raw Materials
IVD Raw Materials
Home » Protein Expression » Customized Bacterial Expression Services » Custom GMP-Grade Bacterial Protein Expression
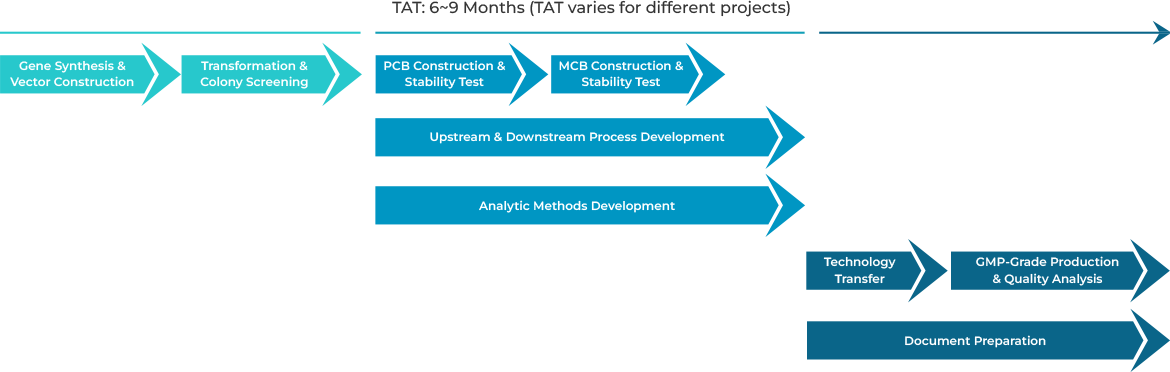
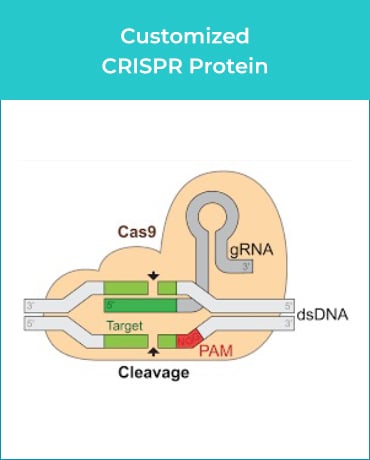
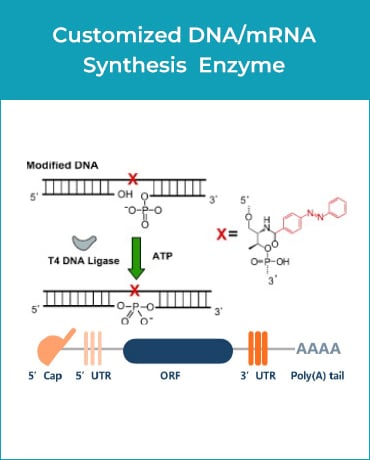
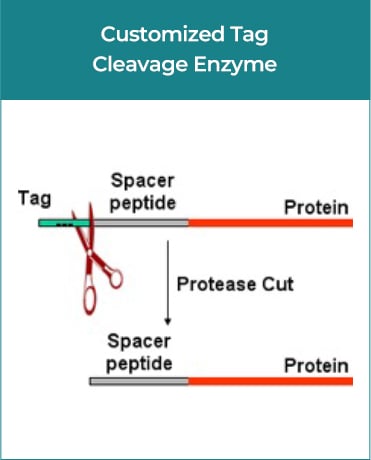
GenScript’s custom GMP-grade bacterial protein expression service produces enzymes under regulatory guidelines for ancillary materials in gene & cell therapy manufacturing processes. Additionally, we offer comprehensive services, including gene synthesis, plasmid construction, cell banking, cell culture, protein purification, formulation, analytic method development, and document preparation.
Delivered by a team with extensive experience in enzyme production processes, our
GMP-grade enzyme service offers several advantages:
1. State-of-the-art facility equipped with advanced instrumentation delivers enzymes in large
quantities and exceptional quality.
2. A highly customizable production process allows parameter optimization based on specific customer
requirements.
3. An adaptable production process from RUO to GMP seamlessly ensures consistent quality throughout
development and manufacturing.
4. The fastest TAT, from gene to protein, enabled by a streamlined production process that delivers
GMP-grade enzymes quickly and efficiently.
5. Extensive documentation, including certificates of analysis and quality control testing results,
supports regulatory compliance and facilitates using GMP-grade enzymes in clinical applications.
Industry-Leading
Enzyme Production
Highly Customized
Production
High Consistency
from RUO to GMP
Fast TAT from
Gene to Protein

Fermentor

Fermentor

High Pressure Homogenizer

Disc Stack Centrifuge

Hollow Fiber Filter

Purification System

Purification System
A: GMP-grade enzymes are manufactured according to guidelines that allow their use as ancillary materials in cell therapy development or other manufacturing processes. GMP proteins are accompanied by extensive documentation and traceability, as well as additional quality control testing.
A: Frequently, the sequence, expression system, and basic manufacturing standard operating procedures (SOPs) for RUO and GMP-grade enzymes are identical, which makes the transition between the two as seamless as possible. However, the key difference between RUO and GMP-grade enzymes lies in their intended use and the level of regulatory oversight applied to their production and testing. RUO enzymes are intended solely for research purposes. They are not subject to the same regulatory scrutiny as GMP-grade enzymes, which are intended for clinical use and must adhere to strict regulatory standards.
A: It is recommended to use GMP-grade enzymes as early as possible in the development process to minimize the risk of contamination and ensure that the final product meets regulatory requirements for clinical use.
A: GenScript's GMP-grade proteins can only serve as vital raw materials in the production of your final product and cannot be used directly in clinical practice or for human injection.
A: GenScript provides COA, datasheet, and TSE/BSE statements at no cost. Please contact us for more details on other documents, such as DMF.
A: Yes, GenScript provides cell-based assays for enzyme in-vivo & in-vitro activity.
A: Yes, all GMP-grade proteins are made in an entirely animal component-free process (ACFP) using E. coli as the source under GMP compliance.

